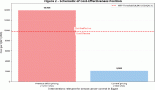
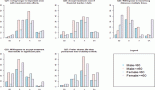
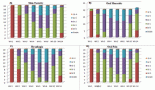
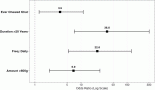
Genitourinary-Rhabdomyosarcomas (GU-RMS) are challenging to treat due to the probable lifelong sequelae of local therapy. Western-world data show 3-year event-free survival (EFS) and overall survival (OS) of 77% and 86%, respectively, for localised disease, with dismal outcomes for metastatic disease. We studied the clinical profile, outcomes and prognostic factors of GU-RMS treated with a multimodal protocol. Treatment-naïve children ≤ 15years with biopsy-proven GU-RMS treated from January 2013 to June 2022 were retrospectively analysed. Local therapy performed at 10–12 weeks of induction was radiotherapy (RT) and/or surgery. Fifty-two patients with a median tumour size of 5.5 cm (range, 3.4–9.2 cm) were analysed. Four patients (7.8%) had alveolar histology. The bladder was the commonest site of primary (36.5%). Group distribution: I-7 (13.4), II-1 (1.9%), III-35 (67.3%) and IV-9 (17.3%). Local therapy was surgery in 11 (21.5%), RT in 25 (49%) or both in 14 (26.9%) patients. With a median follow-up of 56 months (95% confidence interval (CI): 49.1%–63.1%), 4-year EFS for groups I–IV, were 100%, 50% (95% CI: 41%–59%) and 33.3% (95% CI: 2.6%–64%) (p = 0.01), respectively. The corresponding 4-year OS were 100%, 72% (95% CI: 56.4%–87.6%) and 33.3% (95% CI: 2.6%–64%) (p = 0.007), respectively. Relapses were locoregional-4 (7.7%), metastatic-5 (9.6%) and combined-4 (7.7%). Tumour size > 6.45 cm significantly affected outcomes in the localised cohort (hazard ratio = 4.1, 95% CI: 1.38–12.1, p = 0.01). Outcomes of group III GU-RMS in children treated on a multimodal protocol in our study are suboptimal compared to those from co-operative group trials, probably affected by large tumours at presentation, warranting alternative strategies for optimisation of survival.