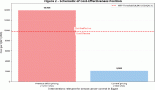
In efforts to modernise the entire drug-development process, making it more efficient, less costly, and ultimately of real benefit to patients, The Federal Drug Administration (FDA) authorised the use of exploratory IND or early Phase I (Phase 0) studies. Quite different in structure from Phase I, II, and III studies, the Phase 0 construct understandably poses a set of ethical problems not seen in the other research phases and so far not adequately addressed by ethicists. In an effort to deal with this deficiency, this paper proposes an ethics critique, based not on the usual concept of benefit, but on the means–end relation, and placed within an ethic of science derived from the practice of science.